What is EDTA?
Contributed by: AeRoGaNiC
Thanks to: aallonharja
(aallonharja)
EDTA-compounds are commonly used in liquid Hydroponic nutrient products to chelate iron and other metallic nutrient ions.
Chelates
Simply put - chelates bond to metals the plant uses for growth, in soil or hydro, and ensures they are stable and mobile. Chelation is an essential for healthy plants. If chelation is poor, the plant will be forced to expend energy to find available nutrients in the medium.
EDTA
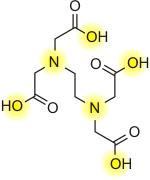 Ethyldiaminetetraacetic acid, also known as EDTA is used to chelate metal ions. EDTA belongs to a class of synthetic compounds known as polyaminocarboxylic acids It has a negative ionic charge, known as an anion, - EDTA4. The important ions are highlighted on yellow. These cations bond with anions to form chelates; ie K+ (potasium anion) will bond with the EDTA cation. Ethyldiaminetetraacetic acid, also known as EDTA is used to chelate metal ions. EDTA belongs to a class of synthetic compounds known as polyaminocarboxylic acids It has a negative ionic charge, known as an anion, - EDTA4. The important ions are highlighted on yellow. These cations bond with anions to form chelates; ie K+ (potasium anion) will bond with the EDTA cation.
The ions from EDTA completely wrap up the entire metal ion all 6 positions. EDTA bonds really well with Magnesium and Calcium as well as many other metal ions like Iron. In fact it appears that Fe is the most finicky element to keep soluble with pH. EDTA is a great stabilizing agent that keeps iron soluble in pH fluctuations. The unusual property of EDTA as a chelating agent is its ability to chelate ( aka complex metal ions) in 1:1 metal to EDTA complexes (If one is familiar with the abilities of chelates, this is a very strong proportion).
Effects on the environment
However, a problem with EDTA is its inability to biodegrade in the environment. EDTA is found in many natural waters and occurs at higher levels in wastewater. This can be a reason to try and avoid EDTA if you can, its inability to dissolve. Western European countries have banned the use of EDTA in detergents, where it is commonly used to chelate metal ions in tapwater. A ban was adopted in Australia as well.
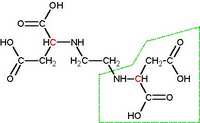 EDDS (S, S'-ethylenediaminedisuccinic acid), a structural isomer of EDTA, has been used as a biodegradable substitute. EDDS is a good complexing agent and is broken down during wastewater treatment processes, unlike EDTA. This is accomplished by performing structural change, which can be seen in the posted pics. The chemical structure of EDDS is organic where EDTA lacks carbon molecules. EDDS (S, S'-ethylenediaminedisuccinic acid), a structural isomer of EDTA, has been used as a biodegradable substitute. EDDS is a good complexing agent and is broken down during wastewater treatment processes, unlike EDTA. This is accomplished by performing structural change, which can be seen in the posted pics. The chemical structure of EDDS is organic where EDTA lacks carbon molecules.
Pic 1: EDTA
Pic 2: EDDS
Organic Chelates
Negative Effects of Synthetic Chelates
EDTA doesnt dissipate in soils and does not break down. This is a serious environmental hazard. Over time EDTA can disserve its function. EDTA can cause reactions in transferable elements and cause precipitation when it fails. One could even argue this stress having an effect on yield, and overall degradation of genetics.
Genetic function relies upon a perfectly balanced equilibrium of environment and chelation. Synthetic chelates can, and often do, interfere with the osmotic equilibrium, by causing precipitation. This imbalance in the environment can cause deficiencies, ergo stress. We want to give our plants the best quality of life. Using organic chelates have demonstrated in a better efficiency of chelation. Organic chelates are just more absorbable and stable than inorganic chelates.
So what the hell can I use?
Humic acid has organic chelates and chelating agents. The microbial activity greatly increases the amount of organic chelating agents being created as well as a natural byproduct. The chelation agents they produce bond with metal ions and become available to plants. Chlorophyll is a chelate that consists of a complex chelating agent with magnesium as the central atom. Hemoglobin in blood is also a great chelating agent. Interestingly enough, the only difference between hemoglobin and chlorophyll is the Fe central atom as opposed to the Mg atom.
Chelation occurs naturally during composting though microbial activity. Adding organic material is also a great way to increase the level of chelating agents by providing beneficial bacteria with food - basically increasing their production of natural chelating agents. Soils with high Cation Exhcange Capacity are generally high in chelating agents and organic content.
What are the benefits?
Theoretically less stress. A smooth chelation process is essential for plants to use minerals and keep them from bonding with each other, this keeps them mobile and stops them from precipitating. Chelating agents nullify the positive charge on the ion and cause it to be more neutral or be a slightly charged anion, encouraging the nutrients to transfer through the pores on the leaf and root surface more rapidly.
Since pores are negatively charged to attract ions, this negative or neutral charge makes ionic bonding and storage of elements in the root pores less likely, increasing the efficiency of the root system.
Sometimes inorganic elements are bound so tightly with their chelates (in synthetic situations) that they cannot be released for physiological function. The plant has a hard time breaking down the inorganic chelates, where as the organic chelates do not share any of the same problems as the synthetics. Using organic chelates has been demonstrated scientifically to produce healthier plants.
One thing is for sure, EDTA is not biodegradable! If you choose to use chemical salts take some time to see what they are chelated with. Even in organics, minerals are sometimes chelated with EDTA, which has been accepted as "organic". However, as I already said it doesn?t break down, and should be avoided if at all possible.
Sources (Chemical Chelates section):
Calculation of EDTA á4 values at any pH
scifun.chem.wisc.edu
Ted Lister, Janet Renshaw :"Understanding Chemistry for Advanced Level"
F. Albert Cotton :"Advanced Inorganic Chemistry"
Sources (Organic Chelates section):
http://www.jhbiotech.com/plant_products/chelation.htm
Bell, Colin F. :"Principles and Applications of Metal Chelation" Oxford, 1977
foliarfert.com/pages/chelates.htm
Tro, Nivaldo J. :"Introductory Chemistry Essentials"
Francis A. Carey, Richard J. Sundberg :"Advanced Organic Chemistry: Structure and Mechanisms"
Richard J. Sundberg, Francis A. Carey :"Advanced Organic Chemistry: Reaction and Synthesis" |
|